- What is SUDEP?
- About CSR
- The CSR Charter
- Participating Centers
- Sponsors & Partners
- Recent Publications
- Recent Funded Projects
“SUDEP refers to the sudden, unexpected death of an individual, with a diagnosis of epilepsy, who dies suddenly, in benign circumstances, without a structural or toxicological cause for death being found at autopsy.”
Awareness is the first step in finding the cause and prevention of SUDEP.
- What causes SUDEP?
- Risk factors for SUDEP?
- What is the incidence of SUDEP?
- Minimizing the risk of SUDEP
The Center for SUDEP Research (CSR) is a National Institute for Neurological Disorders and Stroke (NINDS) funded Center Without Walls for Collaborative Research in the Epilepsies.
This milestone-driven collaboration is composed of researchers from 14 institutions across the United States and Europe and brings together extensive and diverse expertise to understand Sudden Unexpected Death in Epilepsy Patients (SUDEP).
The CSR's scientists and physicians investigate and elucidate these links with their expertise in molecular biology, genetics, histopathology, electrophysiology, brain imaging, data analysis, and more. The team will identify the molecular and structural brain abnormalities underlying SUDEP, utilize these discoveries to identify features that could predict and identify those at risk of SUDEP, and hone in on those targets for the development of potential therapeutic interventions.
Overall, this investment by NINDS over nearly five years promises to catalyze research on SUDEP and dramatically enhance our understanding of this poorly understand and devastating phenomenon.
The CSR brings together leading investigators, clinical and basic science projects, multidisciplinary technology and advanced resources to form a collaborative network dedicated to significantly reducing SUDEP mortality. This spirit of collaboration includes timely sharing of ideas without compromising intellectual autonomy, ownership, and credit..
Members sign the Charter Agreement and agree to a principle of transparency, with particular regard to future grant applications leveraged by CSR, and forthcoming peer reviewed publications, conference presentations, press releases, and other materials arising directly from CSR activities.
A key strategy is to provide each CSR investigator and research group with regular, expedited access to expertise and knowledge as well as core facilities that may not otherwise be available to them.
CSR members are encouraged to share any relevant activities that do not directly arise from CSR resources, cores or scientific projects. Our goal is a broader collaboration within the scientific community to advance knowledge about SUDEP.
This milestone-driven collaboration is composed of researchers from 15 institutions across the United States and Europe and brings together extensive and diverse expertise to understand SUDEP or sudden unexpected death in epilepsy.
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
||||||
The goal of CSR Partners is to foster collaboration and communication between the investigators working within the Center for SUDEP Research and non-profit, academic, industry, and government partners.
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
Featured Publications:
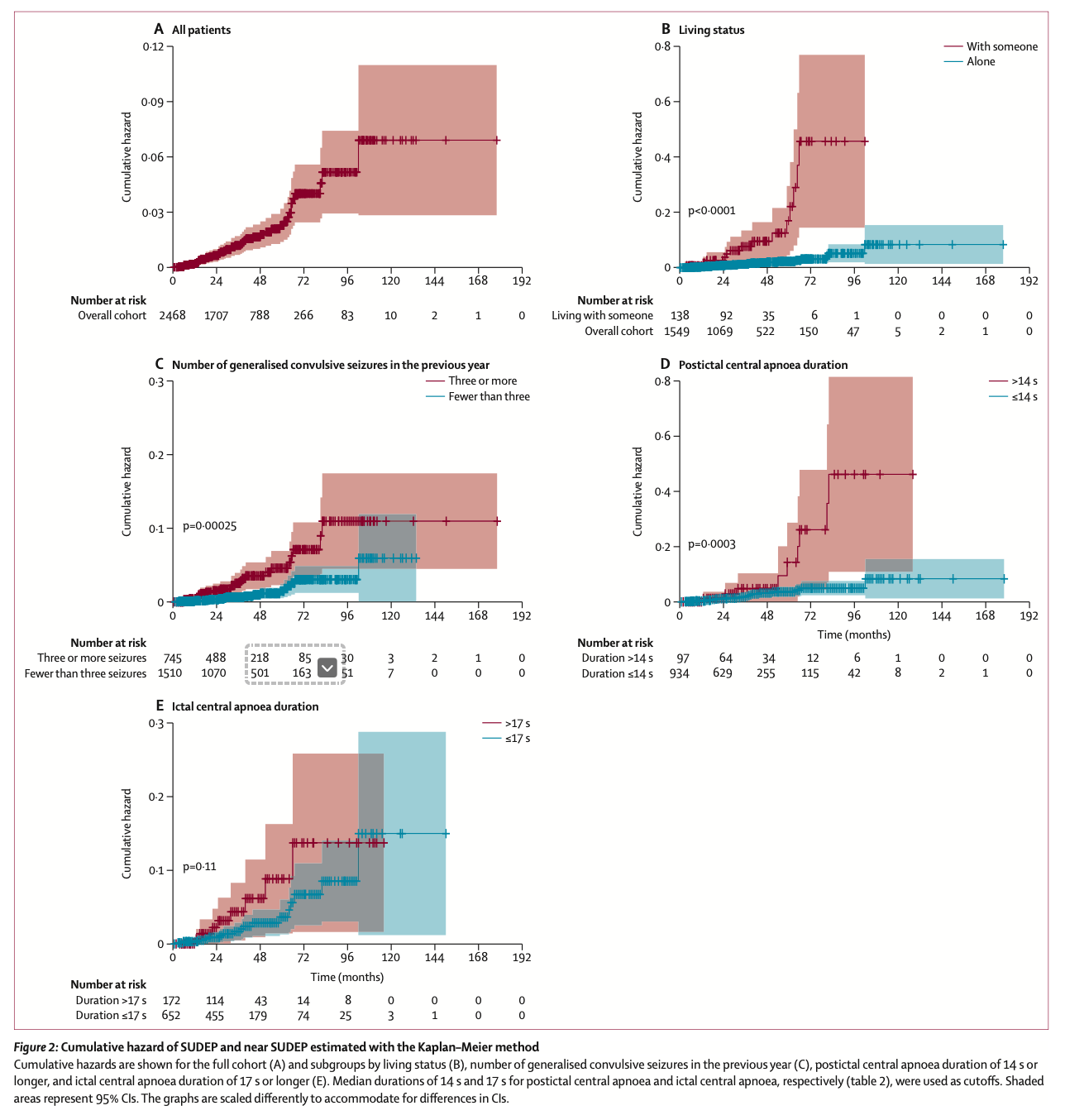
1. The Lancet - Ochoa-Urrea et. al. Risk markers for sudden unexpected death in epilepsy: an observational, prospective, multicentre cohort study. www.thelancet.com Published online September 17, 2025.
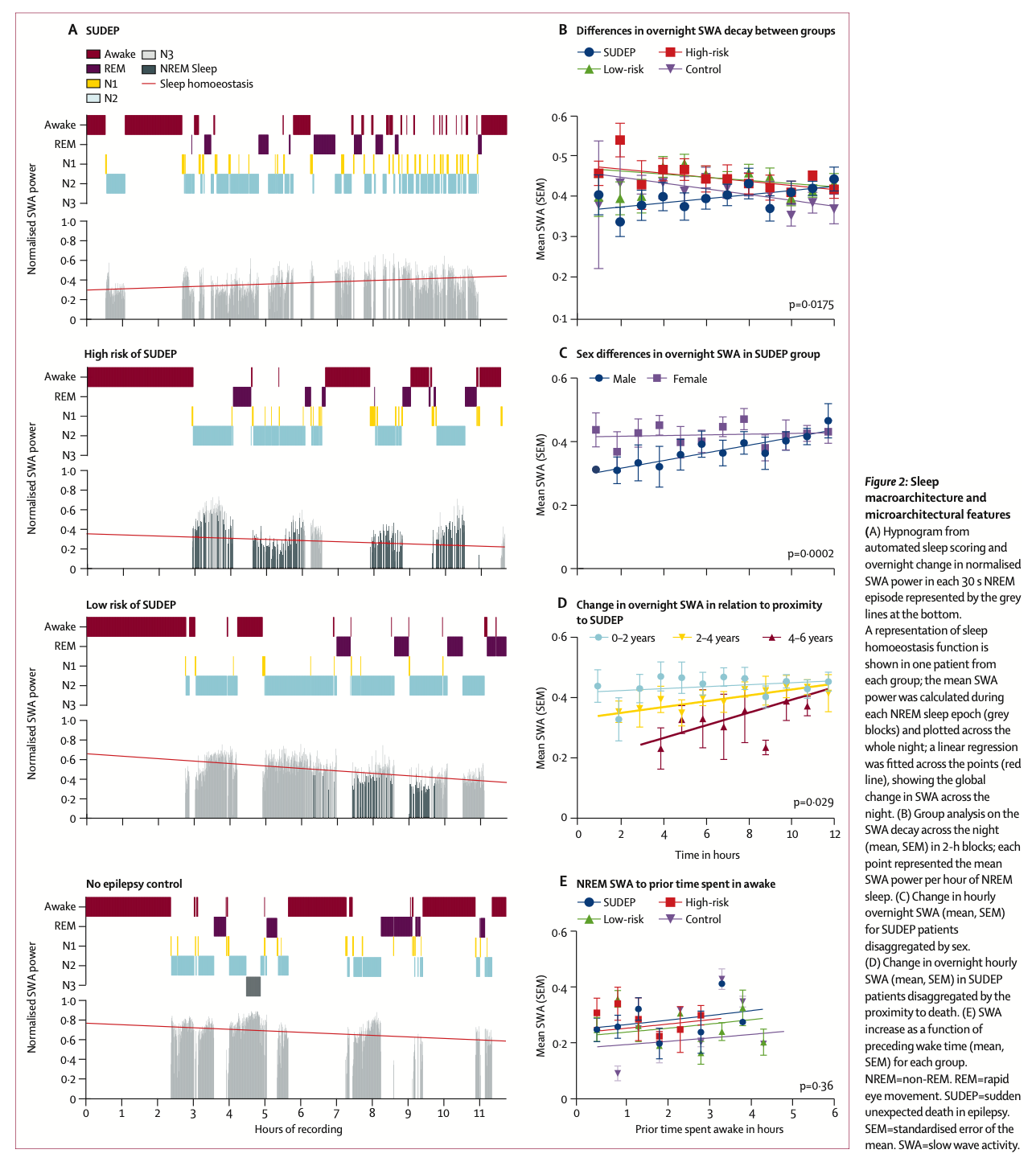
2. The Lancet Neurology - Magana-Tellez et. al. Sleep EEG and respiratory biomarkers of sudden unexpected death in epilepsy (SUDEP): a case–control study. www.thelancet.com/neurology Vol 24 October 2025 840-849.
Recent Funded Projects
International Seizure and SUDEP Research Repository (InSSURR)
PI: Samden D. Lhatoo MD, FRCP; Guo-Qiang Zhang, PhD
Sudden Unexpected Death in Epilepsy (SUDEP) is the commonest category of direct epilepsy related mortality worldwide. Research into biomarker identification and specific prevention approaches has been hindered by the lack of largescale, prospectively acquired, long term (multi-day or longer), multimodality datasets that allow comprehensive study of seizures for clues to mechanisms of death. Understanding mechanisms will allow development of specific preventive methods. The goal of this data resource project, the International Seizure and SUDEP Research Repository (InSSURR), is to overcome the barriers posed by the lack of high-quality datasets for research. InSSURR will be the single largest resource of its kind, expanded from three foundational US and European resources, a) the previously NINDS funded Center for SUDEP Research (CSR) study; b) the French Health Ministry funded European Réseau national d’Etude des facteurs prédictifs et de la Prévention des Morts Soudaines inattendues dans les Epilepsies partielles pharmacorésistantes (REP02MSE) study; and c) the University of Texas at Houston SUDEP research database. InSSURR will ingest seizure and SUDEP data from ongoing and future studies to create the largest publicly available epilepsy mortality research data resource.
SCH: Ensemble Logic: A Formal Precision Phenotyping Framework for Cohort Discovery in Epilepsy
PI: Guo-Qiang Zhang, PhD
In medicine, a phenotype is a person's visible traits or behaviors, such as heart rhythm, brain activity, or signs of disease. Accurately describing these traits is important for making correct diagnoses and choosing the right treatment. But clinicians currently write descriptions of these traits in loose, unstructured text, which can create confusion and inconsistency. Two doctors might describe the same condition in different ways, making it harder to communicate clearly and to keep documented data reliable. This lack of clarity has real effects on health. To diagnose problems like heart or brain disorders, doctors often watch how signals in the body, such as an EKG (electrocardiogram) or EEG (electroencephalogram), change over time. If these changing patterns are not clearly defined, it becomes harder to detect disease early, track its course, or plan treatments tailored to each patient. This project tackles this issue by creating a better way to describe phenotypes. This research builds a new logical system, called Ensemble Logic, that allows clinicians to describe these patterns in a way that is clear, consistent, and easy for computers to understand. This logical system is meant to be both intuitive and flexible. It will be used to define and detect complex health conditions using structured rules that help improve patient care and value in healthcare data.
Breathing Rescue for SUDEP Prevention (BreatheS)
PI: Nuria Lacuey Lecumberri, MD, PhD
Sudden unexpected death in epilepsy (SUDEP) is a devastating complication of epilepsy and was thought to be due to cardiovascular failure, but research on patients who died while being monitored in hospital epilepsy units reveal that most SUDEP is due to post-convulsive central apnea. Crucially, a three-minute post convulsive window of opportunity was identified, beyond which the terminal cascade of respiratory and cardiac failure appears irrevocable. By advancing our understanding of forebrain breathing networks, we can develop neuromodulatory strategies for respiratory facilitation and apnea rescue that may prevent SUDEP during this critical time window. The objective of this project is to understand forebrain modulation of breathing by using anatomically precise intracranial stereotactic electroencephalography (SEEG) data that can determine optimal stimulation paradigms for breathing enhancement.
SCH: Neurophysiological AI-Ready Data Resource
PI: Guo-Qiang Zhang, PhD
The goal of this project is to create an AI-ready resource of semantically annotated collection of electroencephalogram (EEG) recordings and sleep polysomnography data, called the Neurophysiological AI-Ready Data Resource (NAIRD), to transform the access and sharing of such resources for brain health research. NAIRD will leverage data sources from prior repositories that cover two major brain health domains: epilepsy, as a part of the Center for Sudden Unexpected Death in Epilepsy (SUDEP) Research (CSR), and sleep, as a part of the National Sleep Research Resource (NSRR). NAIRD will make this rich collection of electrophysiological signals with associated individual-level health outcomes not only FAIR (Findability, Accessibility, Interoperability, and Reusable), but also interactable and AI-ready.
Advancing SUDEP risk prediction using a multicenter case-control approach
PI: Orrin Devinsky MD; Samden D. Lhatoo MD, FRCP; Daniel Friedman, MD
This study will combine and harmonize data from 86 epilepsy centers to study a retrospective cohort who succumbed to Sudden Unexpected Death in Epilepsy (SUDEP) to identify clinical, electrophysiological and imaging predictors of SUDEP and develop an individualized model using machine learning methods to predict SUDEP risk. In addition to understanding SUDEP mechanisms through studying a large number of SUDEP cases it will also identify the development and validation of an individual risk calculator which could make a big difference with adult patients trying to gain independence.
Cardiac and Autonomic Pathological Markers for Arrhythmias and Sudden Unexpected Death in Epilepsy Patients
PI: David Auerbach PhD
Patients with epilepsy are at high risk of sudden death from SUDEP. We are not able to predict a patient’s risk of SUDEP. Patients without known SUDEP risk factors account for the largest number of SUDEP cases. In contrast to prior risk factor association studies that are not linked to a proposed cause for SUDEP, this study will develop pathological markers for a proposed SUDEP mechanism. Analytics that are validated in the cardiac field, but are new to the epilepsy/SUDEP field will identify the clinical populations at risk of, and the triggers for, cardiac-mediated SUDEP.
Prevention of SUDEP by milk whey: Role of CO2 chemoreception and serotonin
PI: George B. Richerson, MD, PhD
Sudden unexpected death in epilepsy (SUDEP) is the most common cause of death in patients with refractory epilepsy. Emerging data indicate that a substantial percentage of SUDEP is due to seizure-induced respiratory arrest. There is a gap in knowledge about how seizures cause apnea, who is at highest risk and what can be done to prevent it. We have found that postictal death is due to seizure-induced apnea in two genetic mouse models of human SUDEP (Scn1aR1407X and Scn8aN1768D mice). Our data indicate that seizures activate projections from the amygdala to the brainstem causing central apnea, and transient defects in CO2 homeostasis and serotonin (5-HT) neuron function. This is supported by data showing that 5-HT neurons, which are central CO2/pH chemoreceptors that stimulate breathing, are inhibited during seizures. The central hypothesis of the current proposal is that seizures impair CO2 chemoreception, in part by inhibiting 5-HT neurons, which increases the risk of a seizure becoming fatal. We have also found that a diet supplemented with milk whey causes a large reduction in the risk of SUDEP, and this may be due to an increase in 5-HT.
Defining breathing network neuromodulatory approaches for prevention of Sudden Unexpected Death in Epilepsy (SUDEP)
PI: Nuria Lacuey-Lecumberri MD, PhD
Sudden Unexpected Death in Epilepsy (SUDEP) usually occurs in patients with frequent convulsive seizures due to seizure-induced breathing failure. There are currently no technologies or devices available for directly preventing death in patients at high risk. The goal of this project is to improve the overall understanding of breathing control by identifying specific brain areas that are most important for breathing function and stimulation techniques that can be used to prevent seizure-induced breathing failure. The outcome of this research will pave the way for deep brain stimulation devices for breathing rescue as a targeted SUDEP prevention strategy.
An informatics framework for SUDEP risk marker identification and risk assessment
PI: Licong Cui, PhD
The main goal of this project is to develop an informatics approach for automated extraction of SUDEP risk markers from multimodal clinical data to enable individualized SUDEP risk assessment. Success of this study will enable systematic SUDEP risk assessment based on known and putative factors and communication of such risk factors to patients with epilepsy. Ultimately, this study can lead to evidence-based SUDEP risk assessment tools that help clinicians and patients manage potentially modifiable risks, leading to overall reduced SUDEP mortality and improved epilepsy patient care.
Inhibition of central CO2 chemoreception by seizures: Effects on ventilation, cardiovascular control and postictal recovery of consciousness
PI: George B. Richerson, MD, PhD
Sudden unexpected death in epilepsy (SUDEP) is the leading cause of death in patients with refractory epilepsy and is two to four times as common as sudden infant death syndrome (SIDS). Because the mechanisms responsible for SUDEP have not been clearly defined, there are no specific treatments to prevent it. Serotonin (5-HT) neurons are central CO2 chemoreceptors (CCR) that regulate breathing, autonomic function, and arousal. Patients with low interictal CCR are more hypercapnic following a generalized tonic-clonic seizure (GTCS), and seizures may depress CCR, an effect that can be measured with the hypercapnic ventilatory response (HCVR) test. Impairment of CCR by seizures may contribute to autonomic dysfunction and impaired arousal after GTCS, increasing the risk of SUDEP. The long-term goal is to develop new treatments to prevent SUDEP by elucidating the mechanisms responsible for seizure-induced respiratory depression, autonomic dysfunction, and impaired arousal.
The role of central CO2 chemosensitivity in postictal respiratory depression and SUDEP
PI: George B Richerson, MD, PhD
Sudden unexpected death in epilepsy (SUDEP) is the leading cause of death in patients with refractory epilepsy. In many cases, death follows a period of severe seizure-induced respiratory depression, for which the cause is not understood. The proposed research is relevant to public health and the NIH’s mission because a better understanding of the role played by central CO2 chemosensation in the pathogenesis of seizure-induced respiratory depression is expected to lead to the identification of novel candidate biomarkers for SUDEP risk and to the development of new preventive treatments for SUDEP.
Isolating SUDEP risk conferred by genomic co-variation in candidate SUDEP genes
PI: Alica Goldman, MD, PhD
This pilot project aims to evaluate the components of genetic risk in SUDEP in SCN1A gene related epilepsy and in patients with febrile seizure phenotype. Goal of this project is to develop critically needed bioinformatic tools for genomic SUDEP risk stratification. Results will have an immediate impact on patients with epilepsy due to SCN1A gene pathogenic variation and they will inform future bioinformatics and statistical design when analyzing SUDEP risk in patients with the clinically very common febrile seizure phenotype as well as when evaluating causality in existing collections of SUDEP cases.
Latest News
|
CSR |
Sleep EEG and respiratory biomarkers of sudden unexpected death in epilepsy (SUDEP): a case–control study
2025-10-01 |
CSR |
Risk markers for sudden unexpected death in epilepsy: an observational, prospective, multicentre cohort study
2025-09-17 |
|
CSR |
Seizure-induced apnoea and the risk of sudden unexpected death in epilepsy
2025-09-17 |
CSR |
NINDS at the forefront of epilepsy research for 75 years
2025-09-15 |
|
CSR |
Sleep biomarkers for sudden unexpected death in epilepsy
2025-09-15 |
Current Events
|
16-19
Mar
|
Artificial Intelligence in Epilepsy And Neuological Disorders
Mar 16-Mar 19, 2026 PUERTO RICO |